 This is the calculated mass of the object which you can convert into any unit of mass measurement. The formula used by this calculator to determine mass from volume and density is:Įnter volume of the object and select the appropriate volumetric units Density of SubstanceĮnter the known density of the material being measured. A conversion scale for volume versus mass at a fixed density will also be displayed which will relate to each calculated result. 
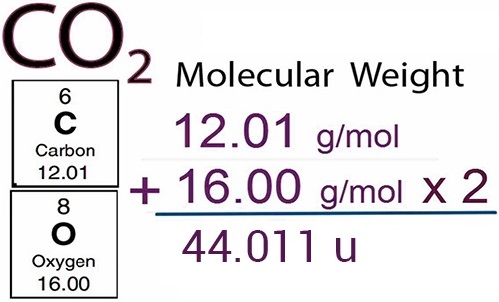
This calculator is used to determine the mass of an object from the measured volume and known density. Determine volume from mass and density measurements.Determine density from mass and volume measurements.Add them together to get the total molar mass of 18.013 g/mol. In this case, the molar mass of the two hydrogen atoms is 2.014 g/mol, while the single oxygen atom is 15.999 g/mol. Example problem: Molar mass of K 39.1 g Molar mass of Mn 54.9 g Molar mass of O 16.0 g (The solute contains 4 O atoms, so count the 16g 4 times. Make sure that you count the atoms for each element and calculate the molar mass of each of the atoms. One atomic mass unit (u) is equal to 1/12 the mass of one atom of carbon-12. Find the molar mass of each element using the periodic table of elements. Don’t forget to take into account the number of atoms of each element when you make your calculation. Molar mass is the mass (in atomic mass units) of one mole of a of a substance. Then, calculate the molar mass of each element in the compound. For example, if you’re calculating the molar mass of water, you’d start with the formula H2O. First, you’ll need to find the chemical formula for the compound. Finding the molar mass of a compound is a little more complicated. For example, the molar mass of H2 is 1.007 x 2 x 1 g/mol, or 2.014 g/mol. In these cases, you’ll need to multiply the relative atomic mass of the element by the number of atoms in the molecule, then multiply the result by the molar mass constant. However, some elements, such as hydrogen, nitrogen, and oxygen, only occur naturally in molecules of 2 or more atoms. For instance, zirconium has a molar mass of 91.22 x 1 g/mol, or 91.22 g/mol. For most elements, this means that the relative atomic mass is equal to the molar mass. Next, multiply the atomic mass by the molar mass constant, which is equal to 1 gram per mole. Our final answer is expressed to four significant figures. The calculated value makes sense because it is almost four times times the mass for 1 mole of aluminum. For instance, the relative atomic mass of zirconium (Zr) is 91.22. Prepare a concept map and use the proper conversion factor. This is usually located under the symbol and name of the element. To find an element’s molar mass, start by checking the periodic table for the relative atomic mass of the element. Molar mass is the mass in grams of 1 mole of any given substance. According to the International System of Units, a mole is the amount of any substance that contains the same number of elementary entities-typically atoms or molecules-as there are atoms in 12 grams of the isotope carbon-12. When you’re measuring extremely tiny amounts of a substance, it can be helpful to use molar mass. One mnemonic device for remembering diatomic elements (molecules of 2 atoms) is: Have No Fear Of Ice Cold Beverages (Hydrogen, Nitrogen, Fluorine, Oxygen, Iodine, Chlorine, Bromine).Multiply them by the molar mass constant, and then multiply the result by 2. This means that if you want to find the molar mass of elements that are composed of 2 atoms, such as hydrogen, oxygen, and chlorine, then you'll have to find their relative atomic masses. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
